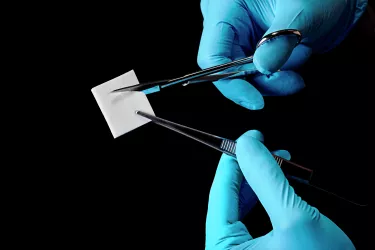
creos™ mucogain: A natural collagen matrix
creos mucogain is a natural collagen matrix for soft-tissue regeneration
Nobel Biocare’s creos regenerative product line offers reliable solutions for guided bone regeneration and guided tissue regeneration. This product range includes a natural resorbable collagen matrix designed to replace autologous graft material in several indications: creos mucogain.
The natural, ready-to-use matrix
Soft tissue management plays an important role in esthetics and function, for the correction of soft tissue recession and volume loss, to promote a natural soft tissue appearance and because soft tissue height can influence early crestal bone loss.1 While autologous soft tissue grafting is a well-established solution, it can present a number of challenges. Harvesting the patient’s soft tissue requires a second surgical site, which is associated with post-operative pain, bleeding, swelling and infection2,3,4 and the palate is a risky surgical site due to the existence of an artery. The quality and quantity of the harvested soft tissue depends upon the patient’s health and may be limited.2,3,4
The soft tissue graft substitute, creos mucogain, is designed to overcome these challenges. A resorbable collagen matrix composed of highly purified porcine collagen and elastin fibers, it is designed to provide a readymade, easy-to-handle alternative to autologous soft tissue grafts.* Its patented production process creates an open interconnecting porous structure,5 designed to promote soft tissue regeneration through the migration of cells and blood vessels into the matrix.6,7
To meet the needs of several indications, such as soft tissue volume augmentation and root coverage in submerged healing,* the matrix provides the clinician with a choice of multiple sizes and thicknesses and for easy handling can be used straight from the box and trimmed to precisely fit the surgical site.8
With strength comes confidence
Creos mucogain puts mechanical strength on display, making fixation with sutures possible thanks to high suture retention both when dry and when hydrated,7 and its suture pull-out strength is sufficient for the tunneling technique.8 High stress resistance also makes creos mucogain easy to handle. The matrix benefits from the memory effect, meaning it retains its initial volume after hydration and cyclic handling simulations in-vitro.7
Ultimately, it is clinical effectiveness that really matters, and in a prospective case series, no significant adverse events related to creos mucogain were observed during the healing phase and up to the last visit.9
References
1 Linkevicius T, Apse P, Grybauskas S, et al. The influence of soft tissue thickness on crestal bone changes around implants: a 1‐year prospective controlled clinical trial. Int J Oral Maxillofac Implants. 2009;24(4):712-719.
Read on PubMed
2 Harris RJ, Miller R, Miller LH, et al. Complications with surgical procedures utilizing connective tissue grafts: a follow-up of 500 consecutively treated cases. Int J Periodontics Restorative Dent 2005;25(5):449-459.
Read on PubMed
3 Griffin TJ, Cheung WS, Zavras AI, et al. Postoperative complications following gingival augmentation procedures. J Periodontol 2006;77(12):2070-2079.
Read on PubMed
4 Aguirre-Zorzano LA, García-De La Fuente AM, Estefanía-Fresco R, et al. Complications of harvesting a connective tissue graft from the palate. A retrospective study and description of a new technique. J Clin Exp Dent 2017;9(12):e1439-e1445.
Read on PubMed
5 Heschel I, et al. 2002. Method for producing porous structures. US patent 6,447,701 B1.
Read online
6 Boekema B, Vlig M, Olde Damink L, et al. Effect of pore size and cross-linking of a novel collagen-elastin dermal substitute on wound healing. J Mater Sci Mater Med 2014;25(2):423-433.
Read on PubMed
7 Damink, LO, Heschel I, Leemhuis H, et al. Soft tissue volume augmentation in the oral cavity with a collagen-based 3D matrix with orientated open pore structure. Current Directions in Biomedical Engineering 2018; 4(1): 237-241. [conference paper].
Read online
8 Wessing B, Vasilic N. Soft tissue augmentation with a new regenerative collagen 3-d matrix with oriented open pores as a potential alternative to autologous connective tissue grafts [329]. Clin Oral Implants Res 2014;25(s10):342. [poster presentation].
Read online
9 Montero E, Sanz-Martin I, Sanz-Sanchez I, et al. Volumetric changes in the buccal contour after immediate implant placement and provisional restoration together with a soft tissue substitute [12687]. Clin Oral Implants Res 2018;29(Suppl 17). [oral presentation].
Read abstract